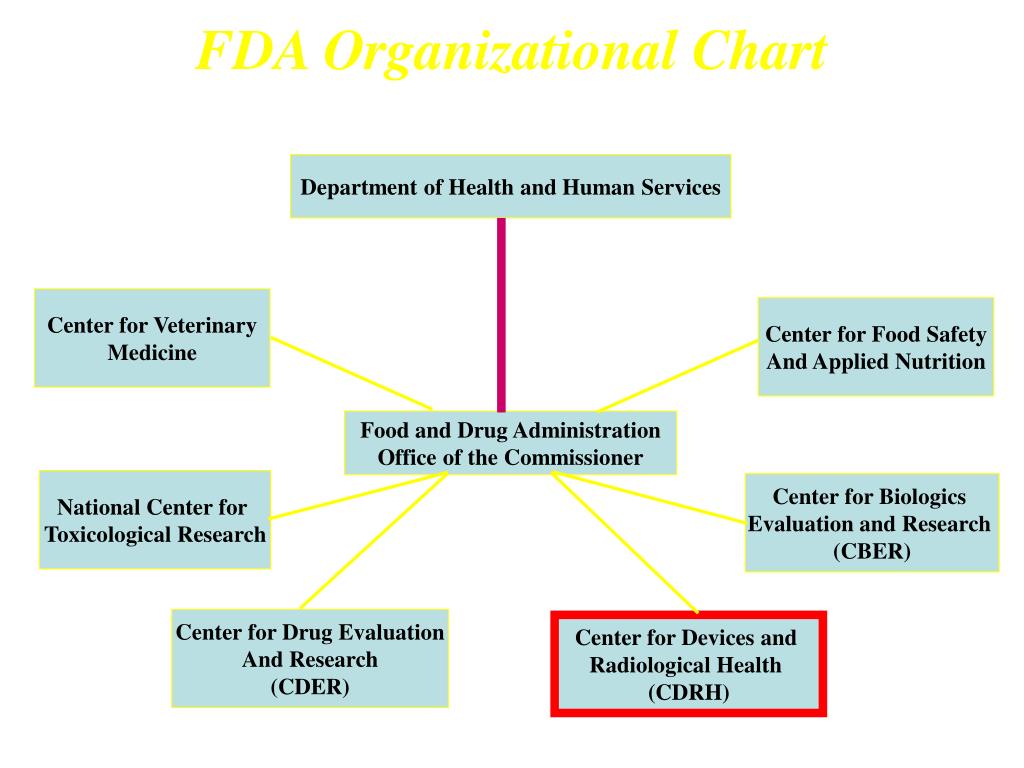
Cdrh Org Chart - Reorganization Of The Center For Devices And Radiological
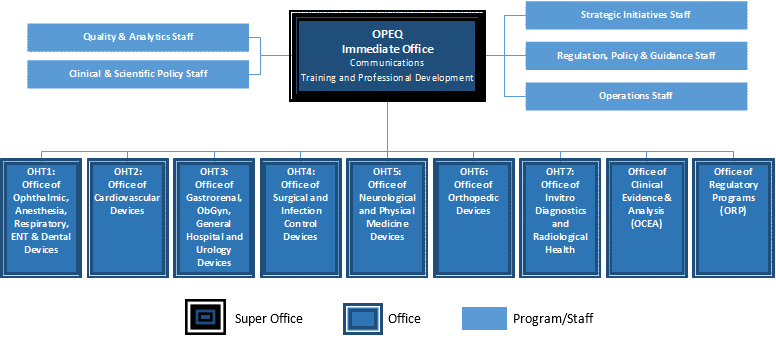
Reorganization Of The Center For Devices And Radiological .
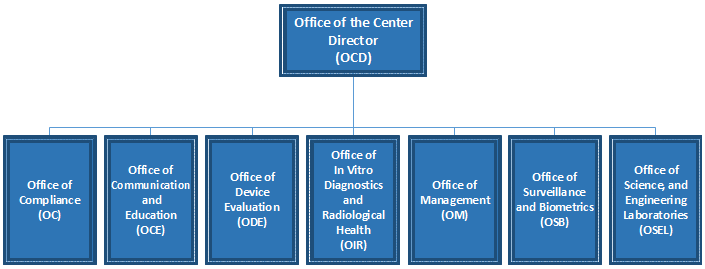
Reorganization Of The Center For Devices And Radiological .
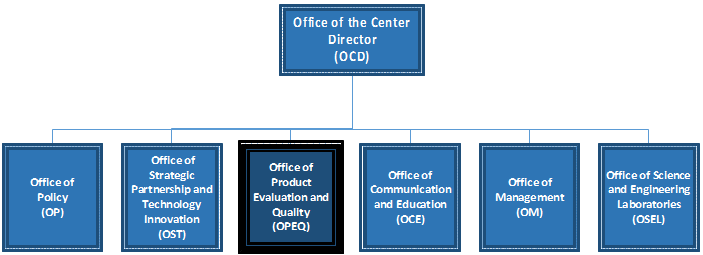
Reorganization Of The Center For Devices And Radiological .

Changes To Fda Laser Enforcement And Inspection Staff Jan 2013 .
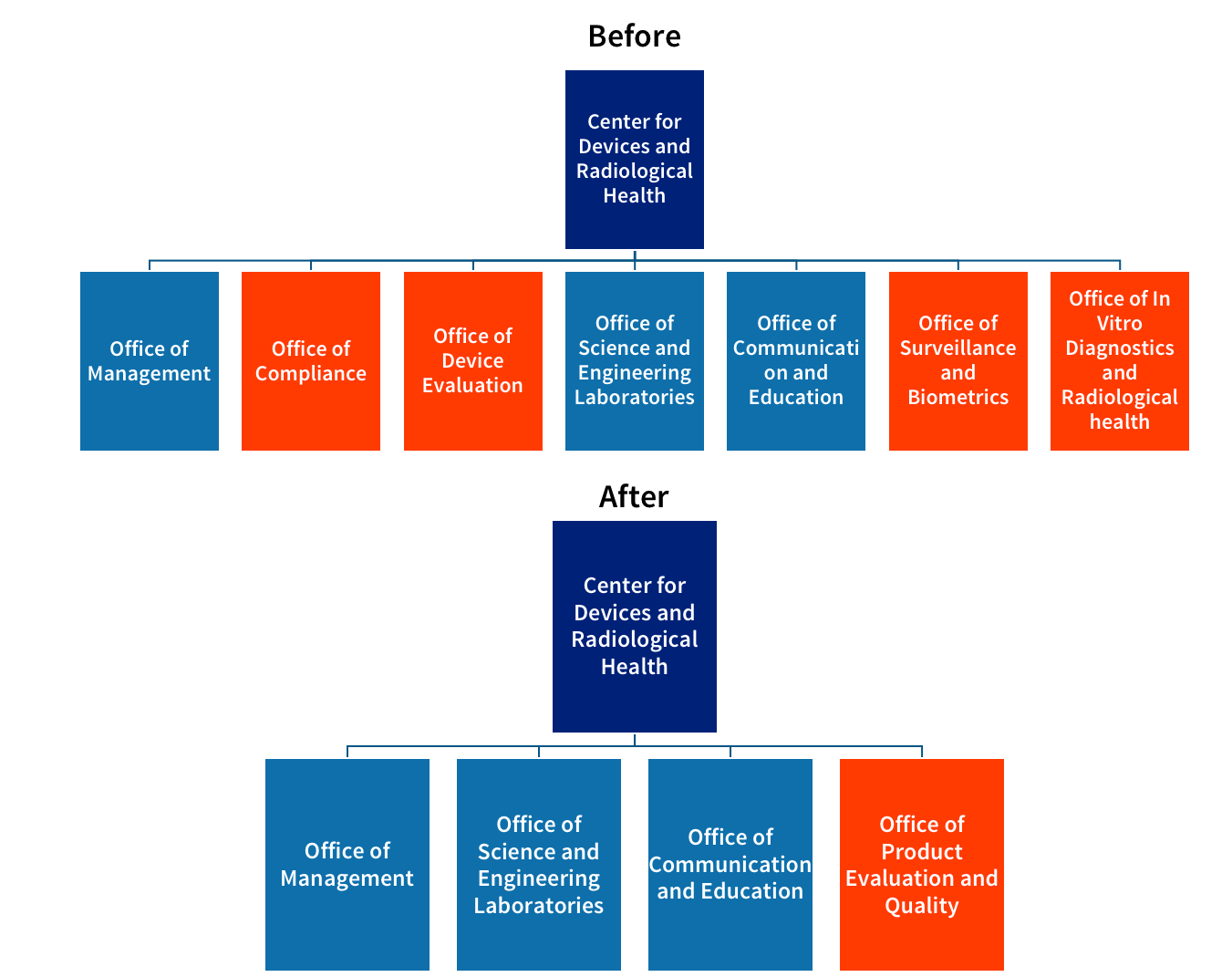
Cdrh Reorganization How Will It Impact Your Medical Device .

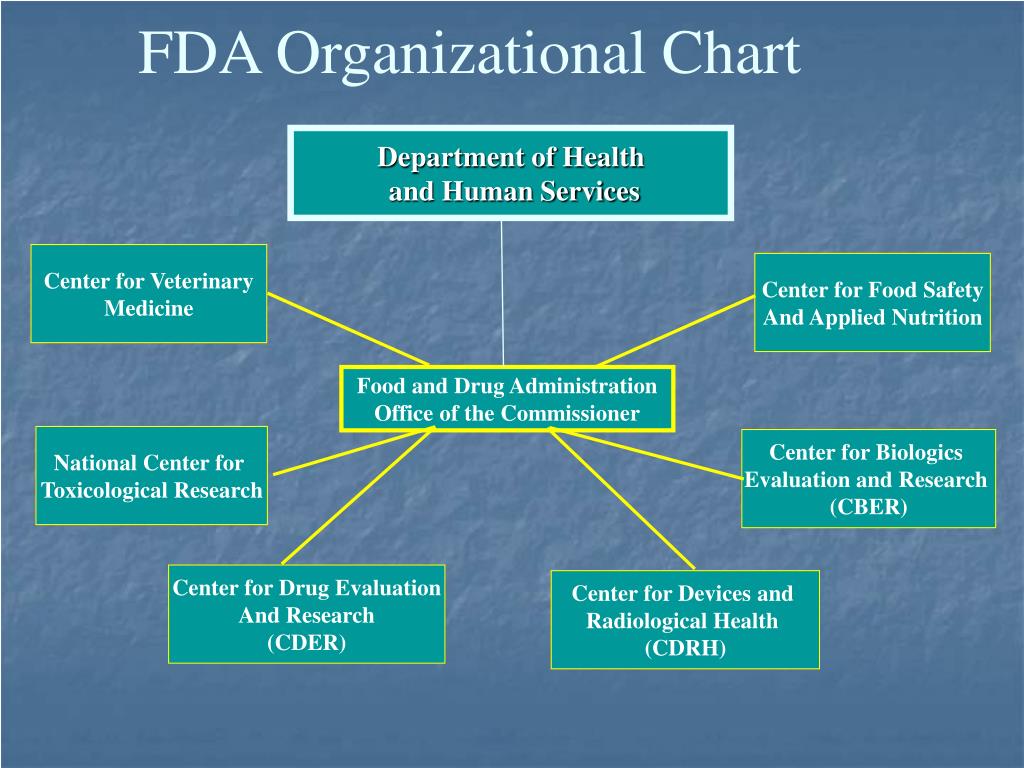
Food And Drug Administration An Overview Sciencedirect .

Reorganization Of The Center For Devices And Radiological .
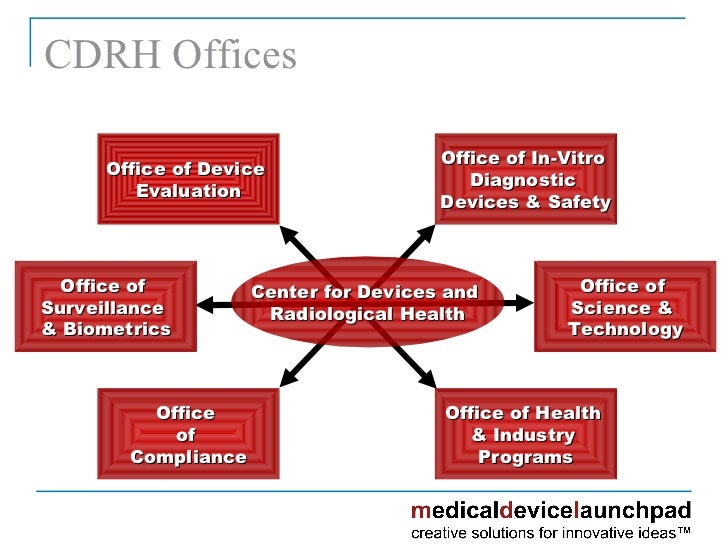
What Is Cdrh Fda .

Entering The Us Market Medical Devices Ppt Video Online .
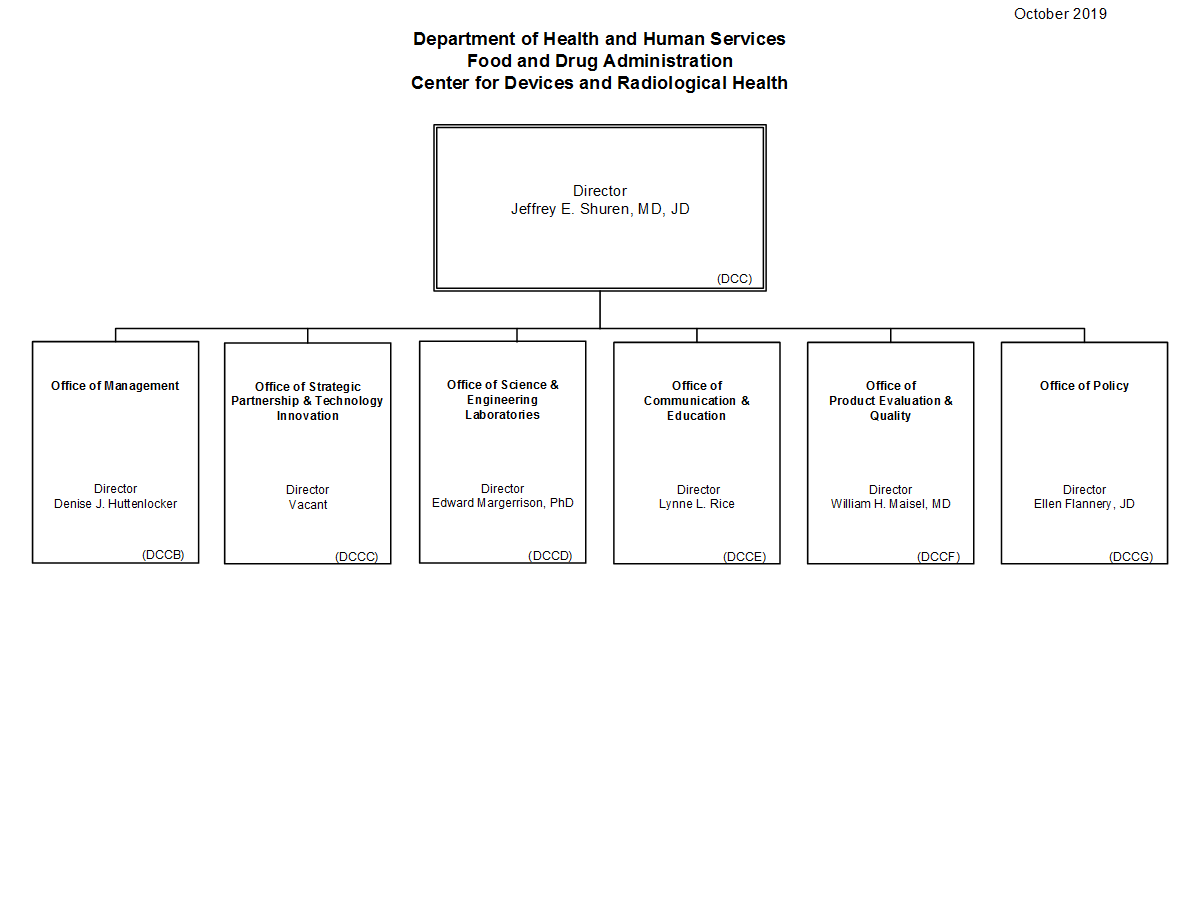
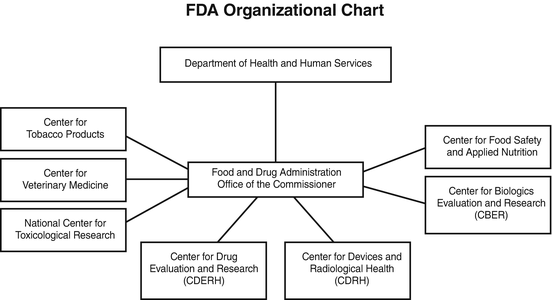
Center For Devices And Radiological Health Organization .

Figure 1 From A Program To Provide Regulatory Support For .
Ppt Use Of Outside Experts In Fdas Premarket Evaluation .
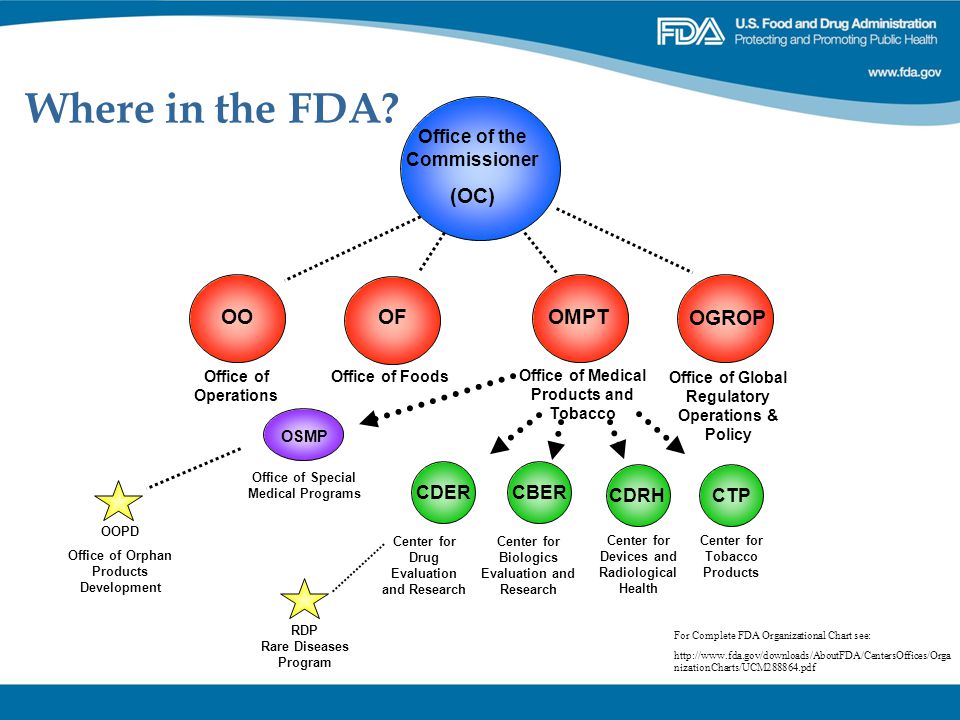
Rare Diseases And Fdasia Ppt Video Online Download .
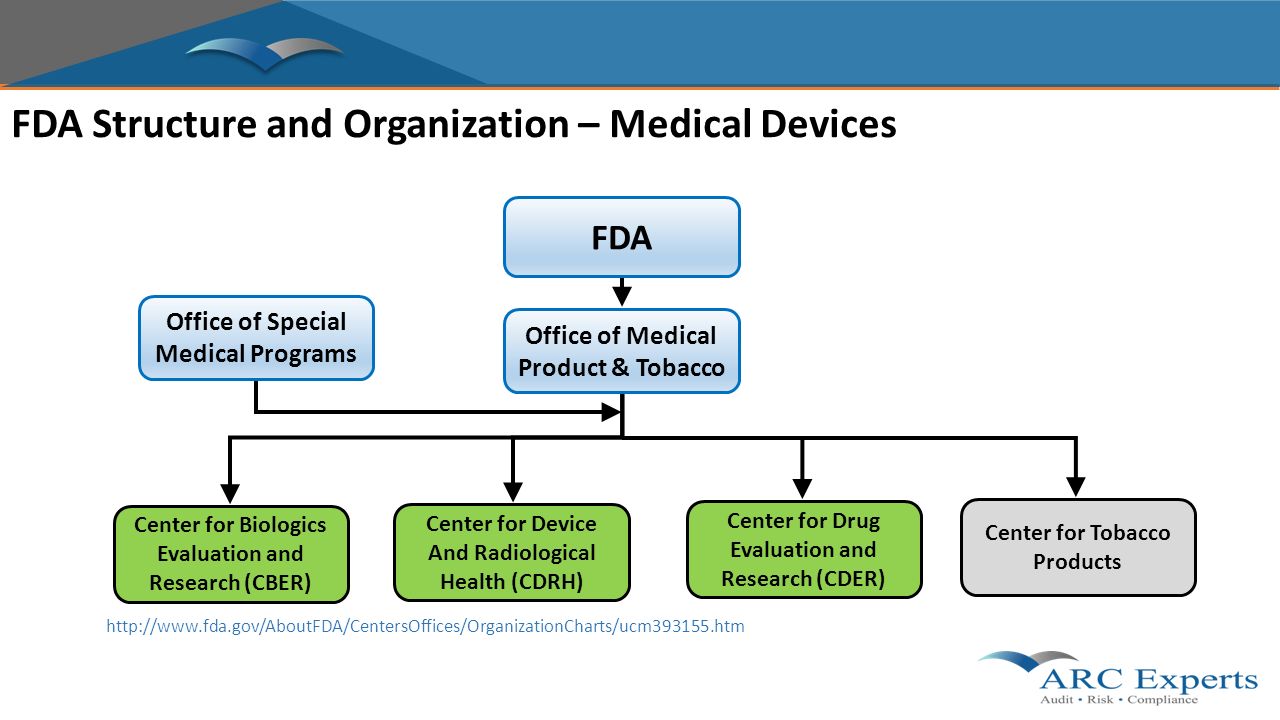
Moj Eram Phd Senior Consultant Arc Experts Llc 6 April .

Figure 2 From Clinical Trials In Orthopaedics Research Part .

21 Eye Catching Fda Cber Org Chart .

Information About Fda Notified Bodies For Medical Device .

Cdrh Mission Center For Devices And Radiological Health Cdrh .

Figure 1 From New Technology In Electrophysiology Fda .
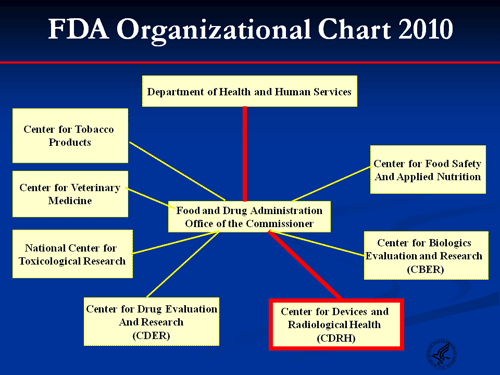
Balancing Innovation Vs Safety And Effectiveness An Fda .
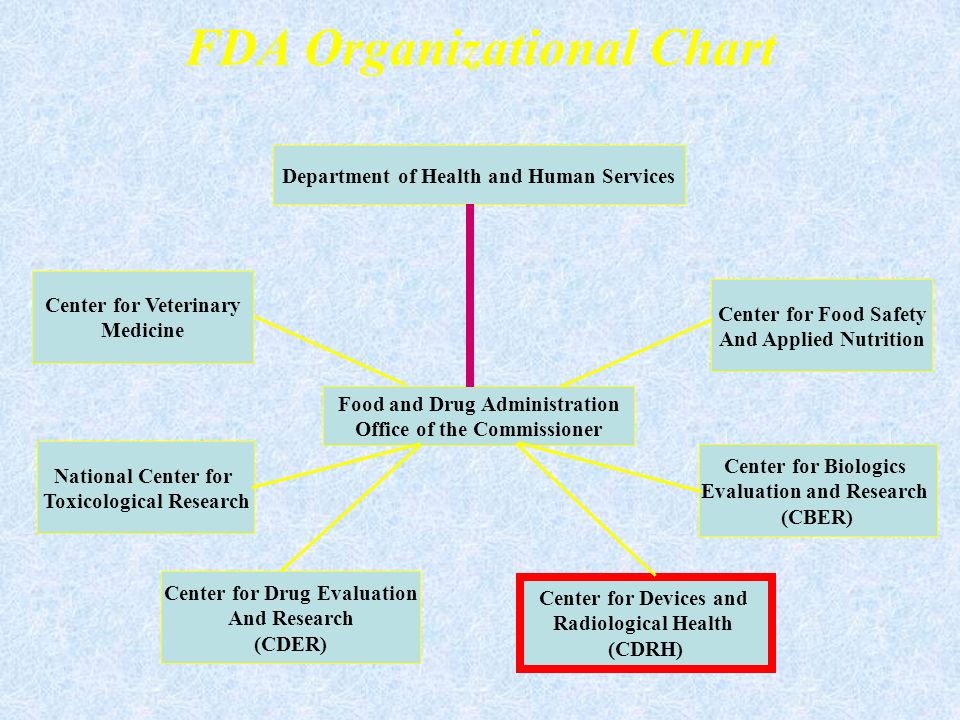
Fda Regulation Of Pharmaceuticals And Devices Department Of .

Information About Fda Notified Bodies For Medical Device .

Cdrh Mission Center For Devices And Radiological Health Cdrh .

Flow Chart From Fda Cdrh Use Of International Standard Iso .

Fda Cdrh New Organizational Structure Effective May 1 2019 .

Ppt Improving Patient Safety By Reporting Problems With .

Harvard Mit Division Of Health Sciences And Technology .

16 Th Annual Nih Sbir Sttr Conference Functional Down To .

Flow Chart From Fda Cdrh Use Of International Standard Iso .

Software Introduction Tools .
Fda Explains What Mobile Apps Are No Longer Devices Raps .
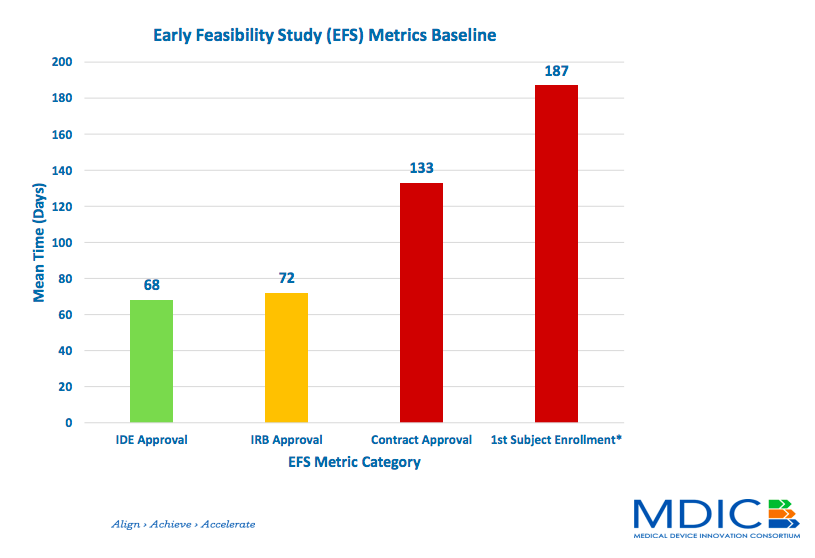
Cdrh Offers Peak Into Plans For Early Feasibility Studies .

Schematic Showing How A Usage Model Is Used To Enhance .

Cdrhs Office Of Compliance Reorganization Rebroadcast .

Preapproval Inspections For Manufacturing Christy Foreman .
Ppt Fda Perspective On Cardiovascular Device Development .

Harvard Mit Division Of Health Sciences And Technology .

Hashtag Fdafail Sur Twitter .

5 Documentation Based On Level Of Concern Cdrh 2005 .

Cber 510 K Issues Sheryl A Kochman Mt Ascp Chief .

Cdrh Mission Center For Devices And Radiological Health Cdrh .

Flow Chart From Fda Cdrh Use Of International Standard Iso .
.png.aspx)
Cdrh Offers Peak Into Plans For Early Feasibility Studies .

Cber 510 K Issues Sheryl A Kochman Mt Ascp Chief .

Quality Management Programs .

Balancing Innovation Vs Safety And Effectiveness An Fda .
Regulatory Issues Springerlink .

Planned Cdrh Reorganization Will Impact Device Approvals And .

New Best Practices For Biosample Management Moving Beyond .

Cdrhs Office Of Compliance Reorganization Rebroadcast .
- jack chart
- horace small pants size chart
- overwatch character counter chart
- sexual exposure chart
- propane pressure chart
- air fryer frozen food cooking chart
- extjs chart legend
- nse live charts with indicators
- psychrometric chart high temperature
- university of michigan seating chart football
- my lahey chart patient portal
- iso 7380 weight chart
- asian gender chart
- capricorn love chart
- primary teeth chart letters
- ashford prep school great chart
- robber baron or captain of industry chart answers
- free printable baby feeding chart
- multiplication chart 3s
- usd to argentine peso chart
- si chart
- c# chart tooltip values
- seadoo fuel consumption chart
- anchor and dmc conversion chart
- bead size chart 11 0
- wave frequency chart
- bass guitar scale chart printable
- summer songs chart
- moa arena seating chart
- malaysian ringgit to gbp chart